ARTÍCULO DE REVISIÓN
Malnutrition and Inflammation—Burning Down the House”: Inflammation as an Adaptive Physiologic Response Versus Self-Destruction?*
La desnutrición y la inflamación—"quemar la casa": La inflamación como una respuesta fisiológica adaptiva o autodestrucción
Gordon L. Jensen, MD, PhD, FASPEN1
1Department of Nutritional Sciences, The Pennsylvania State University, University Park, Pennsylvania.
Gordon L. Jensen, MD, PhD, FASPEN, Department of Nutritional Sciences, Penn State University, 110 Chandlee Laboratory, University Park, PA 16802, USA. Email: GLJ1@psu.edu
*Artículo publicado
en Journal of Parenteral and Enteral Nutrition
Volume 39 Number 1 January 2015 56-62.
© 2014 American Society for Parenteral
and Enteral Nutrition. DOI: 10.1177/0148607114529597
jpen.sagepub.com. Hosted at online.sagepub.com
ABSTRACT
A summary of my 2014 Rhoads Lecture is presented that explores our progress in understanding the complex interplay of malnutrition and inflammation. A historical perspective is provided that highlights the contributions of some of the key pioneers in the nutrition assessment field. Advances in agriculture, education, public health, healthcare, and living standards have affected traditional settings for malnutrition. The chronic disease, surgery, and injury conditions that are associated with modern healthcare are becoming prevalent settings for malnutrition. One consequence has been a growing appreciation for the contributions of inflammation to malnutrition in these clinical conditions. This recognition has driven a fresh look at how we define and think about malnutrition syndromes. An inflammatory component is included in the definitions suggested by the recent Academy of Nutrition and Dietetics and American Society for Parenteral and Enteral Nutrition consensus report that also describes characteristics recommended for the identification and documentation of malnutrition. Efforts are currently underway to evaluate the feasibility and validity of this approach. Recent advances in research highlight the profound impact of inflammation-mediated erosion of muscle mass on clinical outcomes. Research to identify better biomarkers of inflammation and malnutrition must be a leading priority. New “omics” approaches are an especially promising avenue of biomarker investigation. Inflammation can be a good thing; let’s try to keep it that way. (JPEN J Parenter Enteral Nutr. 2015;39:56-62)
Keywords: malnutrition; inflammation; assessment; diagnosis.
RESUMEN
Se presenta un resumen de mi Conferencia de Rhoads
2014 que explora nuestro progreso en la comprensión de la compleja interacción
de la desnutrición y la inflamación. Se proporciona una perspectiva
histórica que pone de relieve las contribuciones de algunos de los principales
pioneros en el campo de la evaluación nutricional. Los avances en la
agricultura, la educación, la salud pública, la asistencia sanitaria,
y los niveles de vida han afectado a la configuración tradicional de
la desnutrición. Las condiciones crónicas de la enfermedad, cirugía
y lesiones que están asociadas con la atención sanitaria moderna
son cada vez más la configuración prevalente de la desnutrición.
Una consecuencia ha sido la creciente evaluación de las contribuciones
de la inflamación a la desnutrición en esta situación clínica.
Este reconocimiento ha impulsado una nueva mirada a cómo definimos y
pensamos en los síndromes de mala nutrición. Está incluido
un componente inflamatorio en las definiciones propuestas en el informe de consenso
de la reciente Academia de Nutrición y Dietética y la Sociedad
Americana de Nutrición Parenteral y Enteral que también describe
las características recomendadas para la identificación y documentación
de la desnutrición. Actualmente se está trabajando para evaluar
la viabilidad y la validez de este enfoque. Los recientes avances en la investigación
ponen de manifiesto el profundo impacto de la erosión mediada por la
inflamación de la masa muscular en los resultados clínicos. La
investigación para identificar mejores marcadores biológicos de
la inflamación y la desnutrición debe ser una de las principales
prioridades. Los nuevos enfoques "ómicos" son una vía
especialmente prometedora de investigación de los biomarcadores. La inflamación
puede ser una buena cosa; vamos a tratar de que lo siga siendo. (JPEN J Parenter
Enteral Nutr. 2015;39:56-62)
Palabras clave: desnutrición; inflamación; evaluación; diagnóstico.
Introduction
I want to thank American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) president Ainsley Malone, the A.S.P.E.N. Board of Directors, the A.S.P.E.N. membership, and the legacy of Jonathan Rhoads for the distinct privilege of sharing my observations in the 2014 Rhoads Lecture. Much has transpired since 2006 when I gave my presidential address at Clinical Nutrition Week in Dallas, Texas, which focused on inflammation as the key interface of medicine and nutrition and highlighted opportunities to bring nutrition to the forefront of patient care.1 There is a growing appreciation among nutrition practitioners for the contributions of inflammation in the development of malnutrition in the settings of disease, surgery, and injury. Between 1996 and 2006 there were some 800 PubMed citations with keywords malnutrition and inflammation, and in the 8 years since there have already been more than twice that number. New consensus definitions and characteristics of malnutrition syndromes that incorporate an inflammatory component have been proposed and are currently being tested and evaluated. The summary of my Rhoads Lecture that follows examines where we have been, where we are now, and where we are going. A historical perspective is critical to understanding our current trajectory and to appreciate the opportunities that lie ahead. Many important questions remain for nutrition investigators. Unless otherwise specified, in this paper I use the term malnutrition to refer to undernutrition.
Pioneers
Jonathan Rhoads was the son of a Quaker physician. He attended Haverford College and completed his MD at Johns Hopkins University School of Medicine. He died January 3, 2002, at the age of 94 years after spending almost 70 years affiliated with the Hospital of the University of Pennsylvania. He began his internship there in 1932 and later served as the chair of surgery and provost. A renowned researcher, scholar, and medical leader, he is widely acknowledged as one of the pioneers of nutrition support. He collaborated with Drs Stanley Dudrick, Douglas Wilmore, and Harry Vars on landmark parenteral nutrition studies in dogs and then humans.2 I had the distinct pleasure of having dinner with Dr Rhoads some years back; a remarkable man, he shared incredible stories of his early research efforts that were inspirational in my own career. It is fitting that A.S.P.E.N.’s most preeminent award carries Dr Rhoads’ name.
Many others have contributed to our understanding of malnutrition. The 1970s represented a watershed decade of progress. John Waterlow published his groundbreaking “Classification and Definition of Protein-Calorie Malnutrition” in 1972.3 Bruce Bistrian and George Blackburn reported the prevalence of malnutrition in general surgical (1974)4 and medical patients (1976)5 in 2 of the most highly cited nutrition papers published in our field. Charles Butterworth contributed a sentinel call to action in his 1974 paper “The Skeleton in the Hospital Closet.”6 In the 1980s, Khursheed Jeejeebhoy and colleagues developed Subjective Global Assessment, not only the gold standard of currently available nutrition assessment tools but also one of the first to incorporate a metabolic stress of disease component.7,8 The latter component is a proxy indicator of inflammation.
Malnutrition: An Evolving Story
Historically, famine and starvation were prevalent causes of malnutrition. These are still prominent causes of malnutrition in developing countries. Poverty and food insecurity remain worldwide concerns. Natural disasters like earthquakes and tsunamis continue to compromise food availability. Environmental issues including global warming and drought raise ongoing concerns for agriculture and food supply. Conflict, war, and genocide interrupt availability of food, and food deprivation is often used as a weapon. Indeed, the numbers of refugees and displaced populations likely to be at nutrition risk have increased from 1964 to 2011.9
With improvements in agriculture, education, public health, healthcare, and living standards, the settings for malnutrition have evolved over the past century. People are living longer, and malnutrition in the settings of disease, surgery, and injury has become common. Malnutrition now encompasses the full spectrum of undernutrition and overnutrition (obesity). In many developed countries, obesity is now a far greater public health concern than undernutrition in relation to prevalence, morbidity, and cost. It must also be highlighted, however, that undernutrition can also be found among obese persons who consume poor-quality diets and among hospitalized obese persons with disease, surgery, or injury.10-12 Undernutrition may also contribute to the sarcopenic obesity that is now common among obese older persons.13-16 A recent report from the Overseas Development Institute in the United Kingdom found that the number of adults who are obese or overweight in the developing world more than tripled between 1980 and 2008. 17 This observation means that some developing countries now face a dual burden of prevalent classic malnutrition as well as increasing obesity.
Historic definitions for malnutrition syndromes suffer limitations that include the use of diagnostic criteria that lack full validity, resulting in poor specificity, sensitivity, and interobserver reliability.18 Definitions overlap, and misdiagnosis is frequent. Not only do physicians and other healthcare practi-tioners often receive little training in assessment and diagnosis of malnutrition, but multiple definitions of malnutrition syndromes are found in the literature, promoting widespread confusion. The scope of the problem is illustrated by the frequency by which malnourished patients in the hospital are not diagnosed as such. Corkins and colleagues19 examined International Classification of Diseases, Ninth Revision (ICD-9) malnutrition code diagnoses from the 2010 Healthcare Cost and Utilization Project (HCUP), which contains discharge data from 1051 hospitals in 45 U.S. states.19 The investigators found that only 3.2% of hospital discharges had a malnutrition diagnosis. Those patients who had a malnutrition diagnosis were older, had longer lengths of stay, and incurred greater costs. They were also more likely to have comorbidity, to be discharged to home care, and to suffer mortality. The limitations of this very thorough analysis highlight many of our present difficulties. Prevalence rates for malnutrition in the hospital setting are typically reported in the 30%–50% range depending upon the patient population studied and the criteria used, 4,5,20-22 so many malnourished patients in HCUP were likely not so coded. Patients who were coded as malnourished may or may not have actually been malnourished. The adverse outcomes associated with a malnutrition diagnosis in this analysis may well have been affected by other covariates. Malnutrition often keeps bad company, like serious underlying diseases or conditions, that may contribute to adverse outcomes. While 13.4% of the HCUP patients who received a malnutrition diagnosis received either parenteral or enteral nutrition, it is not possible to address the benefit of these interventions in this type of retrospective healthcare database analysis.
Building Consensus
The growing appreciation for the contributions of inflammation to malnutrition in clinical settings has driven a fresh look at these fundamental issues. In 1997, Roubenoff et al.23 published “Standardization of Nomenclature of Body Composition in Weight Loss,” a call for development of consensus definitions for malnutrition syndromes. This seminal paper included characterization of cachexia as a malnutrition syndrome with underlying inflammation. Multiple initiatives have since attempted to build a consensus definition for cachexia, and an inflammatory component is now broadly accepted.24-27 For example, the European Society for Clinical Nutrition and Metabolism characterized cachexia as a systemic proinflammatory process that is associated with metabolic derangements that include insulin resistance, increased lipolysis, increased lipid oxidation, increased protein turnover, and loss of body fat and muscle.24 This syndrome of anorexia, weight loss, and metabolic alterations is associated with persistent cytokine-mediated inflammation of mild to moderate intensity unless the underlying condition is successfully addressed.24
Sarcopenia has also attracted considerable attention in consensus-building efforts. It was originally defined as loss of muscle mass associated with aging,28,29 but more recent findings suggest greater complexity because changes in strength do not correlate strongly with changes in muscle size. 30,31 Therefore, newer consensus definitions include a functional component. For example, the International
Working Group on Sarcopenia32 defined sarcopenia as poor functioning plus reduced ratio of appendicular lean mass/(height),2 and the European Working Group33 defined sarcopenia as low muscle mass plus reduced gait speed or low muscle strength with normal gait speed. It has been suggested that smoldering subclinical inflammation may contribute to the development of sarcopenia as the low-grade inflammatory state of aging may be associated with reactive oxygen species and increased levels of tumor necrosis factor (TNF), interleukin (IL)-1, IL-6, C-reactive protein, and fibrinogen. 34 Sarcopenic obesity is manifest in many obese older persons as decreased muscle mass and function.13-16 Inflammation- promoted erosion of muscle mass as well as a vicious cycle of progressive physical inactivity with increased adiposity and accumulated disease burden likely culminates in sarcopenic obesity.15 A dire need to establish consensus is highlighted by a recent analysis by Batsis and colleagues. 35 Eight different definitions for sarcopenic obesity were identified by systematic literature review and then applied to 4984 persons aged ≥60 years from the National Health and Nutrition Examination Survey 1999–2004 who completed dual-energy absorptiometry for body composition. The prevalence of sarcopenic obesity varied from 4.4% to 84.0% for males and from 3.6% to 94.0% for females using the different definitions. The overall prevalence varied an incredible 26-fold depending on the definition used.
In a 2009 commentary aptly titled “Malnutrition Syndromes: A Conundrum vs Continuum,”18 I collaborated with colleagues Bruce Bistrian, Ronenn Roubenoff, and Douglas Heimburger to critically revisit the historic definitions for malnutrition syndromes. An updated approach that incorporated a current understanding of systemic inflammatory response was proposed to redefine the syndromes of marasmus, cachexia, and protein-calorie malnutrition. This construct was extended by an International Consensus Guideline Committee convened under the auspices of A.S.P.E.N. and the European Society for Clinical Nutrition and Metabolism that provided further insights regarding inflammation and malnutrition. This paper was co-published in JPEN and Clinical Nutrition in 201037,38 and included the following observations:
· “The pathophysiology of malnutrition that is associated with disease or injury invariably consists of a combination of varying degrees of under- or over-nutrition and acute or chronic inflammation, leading to altered body composition and diminished biological function.”36-38
· “The point at which the severity or persistence of inflammation results in a decrease in lean body mass associated with functional impairment would be considered disease-related malnutrition.”36-38
Inflammation promotes malnutrition and adverse outcomes through associated anorexia and decreased food intake as well as altered metabolism with elevation of resting energy expenditure and increased muscle catabolism. Derangements of micronutrient status including vitamin D, iron, and zinc are also well described.39-46 Inflammation blunts favorable response to nutrition interventions, and the associated malnutrition may render medical therapies less effective.
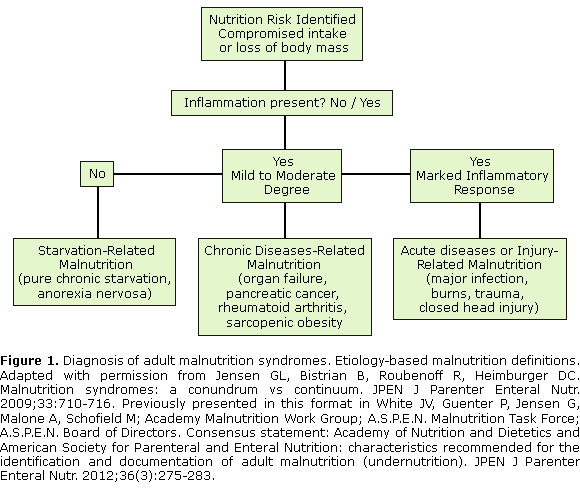
The International Consensus Guideline Committee proposed an etiology-based construct for adult malnutrition in the clinical practice setting. 37,38 The proposed syndromes include starvation-associated malnutrition, when there is chronic starvation without inflammation (eg, anorexia nervosa or major depression with lack of interest in eating); chronic disease-associated malnutrition, when inflammation is chronic and of mild to moderate degree (eg, organ failure, pancreatic cancer, or sarcopenic obesity); and acute disease or injury-associated malnutrition, when inflammation is acute and of severe degree (eg, major infection, burns, trauma, or closed head injury) (Figure 1).37,38 The Academy of Nutrition and Dietetics and A.S.P.E.N. have since extended this approach with a consensus document and new nomenclature for the corresponding syndromes that include malnutrition in the context of social or environmental circumstances, malnutrition in the context of chronic illness, and malnutrition in the context of acute illness or injury.47,48 Clinical characteristics are proposed for the identification and documentation of these malnutrition syndromes in adults that include energy intake, weight loss, and physical findings (subcutaneous fat, muscle mass, fluid retention, and reduced grip strength).47,48
A systematic approach to adult nutrition assessment that supports the new diagnostic construct from the Academy and A.S.P.E.N. was recently proposed that incorporates history and physical diagnosis, clinical signs and physical examination, anthropometric data, laboratory indicators, dietary assessment, and functional outcomes.49
Efforts are currently underway to evaluate the feasibility and validity of the Academy–A.S.P.E.N. malnutrition consensus recommended clinical characteristics. The feasibility of accessing data to support this approach was investigated in 2 tertiary teaching hospitals among 262 consecutive adults referred for nutrition assessment.50 Data were available for food intake history (76% of assessments), weight history (67%), examination for loss of muscle and subcutaneous fat (94%), and examination for edema (84%). Handgrip strength was not available. Decline in food intake and weight loss were the most commonly used characteristics for diagnosis of malnutrition. It was concluded that data were generally available to support diagnosis using the new clinical characteristics and that training in assessment methods and dissemination of necessary tools will be needed. A collaborative multisite validation study has been initiated by the Academy and A.S.P.E.N. Participating dietitians will receive training in nutrition-focused physical examination at Cleveland Clinic. Measures will include clinical characteristics, laboratory results, vital signs, infections, life quality questionnaire, and subjective global assessment. Outcomes will include length of stay, falls, pressure wounds, infections, readmissions, emergency department visits, mortality, and coding of dis-charge diagnoses.
Another exciting development is the recent extension of the etiology-related approach that incorporates inflammation to define pediatric malnutrition syndromes. Mehta and colleagues51 noted that “in the developed world, malnutrition is predominately related to disease, chronic conditions, trauma, burns or surgery.” Their proposed construct for defining malnutrition in hospitalized children corresponds to that described above for adults, but a key distinction is the necessity of addressing development and growth outcomes.
New Inflammation Findings
It is well established that a host of medical conditions are associated with inflammation.1 This line of inquiry has now been extended to the effects of emotional stress. Greater risks of depression, cardiovascular disease, diabetes mellitus, autoimmune diseases, upper respiratory infections, and poor wound healing have been described in persons with emotional stress.52
Bereavement is associated with increased morbidity and mor- tality in the surviving spouse.53 Indeed, higher circulating lev- els of IL-1RA and IL-6 have been detected in grieving individuals.53 Among 276 adults exposed to rhinoviruses and followed for 5 days for cold symptoms and viral shedding, those with recent exposure to stress exhibited glucocorticoid receptor resistance and were at greater risk to develop a cold.54
A subset of 79 adults who were followed for 5 days after rhino- virus exposure underwent measurement of cytokines in their nasal secretions.54 Greater glucocorticoid receptor resistance was associated with higher levels of proinflammatory cytokines.
The importance of muscle in inflammatory states is illustrated by an innovative investigation from Vickie Baracos at the University of Alberta and her collaborators at M. D. Anderson Cancer Center.55 Lumbar skeletal muscle index (skeletal muscle area in cm2 at L3/height in m 2) imaged by computed tomography was predictive of survival in 104 patients with advanced cancer. Of particular interest, 63% of these patients had body mass index ≥25 kg/m2, and reduced muscle was associated with decreased survival irrespective of body size.
In my 2006 presidential address I noted that nutrition supplementation alone is ineffective in preventing muscle protein loss in the presence of significant inflammation.1 This provocative assertion met with some consternation among the audience members at that time. A recent publication by Puthucheary and colleagues56 addressed this issue in a most elegant fashion. These investigators recruited 63 patients, within 24 hours of intensive care unit admission, with a mean Acute Physiology and Chronic Health Evaluation II (APACHE II) score of 23.5 including 49% with sepsis and 25% with trauma. Serial muscle ultrasound for cross-sectional area and biopsy for histopathology and ratio of protein to DNA were obtained. Muscle protein synthesis was measured by leucine incorporation into the vastus lateralis using constant infusions of [1,2 13C2] leucine on days 1 and 7, and leg protein breakdown and balance were simultaneously measured by femoral vein dilution of D5 phe-early onset of systemic inflammatory response syndrome innylalanine during a primed constant infusion. Muscle loss occurred early and rapidly during the first week despite the administration of nutrition support. It was even more pronounced in those with multiorgan failure. Cross-sectional muscle area declined over 7 days as did the ratio of protein to DNA. Protein synthesis as determined by fractional synthetic rate was depressed at day 1 compared with fasted healthy controls and increased by day 7 to rates observed with fed controls. Leg protein breakdown remained elevated throughout the study such that the net balance remained catabolic. Histopathological testing revealed myofiber necrosis and a cellular infiltrate of macrophages by day 7 as well.
Intervention Issues
It is important to recognize that anti-inflammatory interven- tions often have undesirable side effects—steroids or anti- TNF, for example. Many anti-inflammatory interventions also blunt desirable aspects of inflammation. Raising the white blood cell count, mounting a fever, synthesizing needed acute reactants for immune response and wound healing, and breaking down muscle to provide substrate are all potentially adaptive physiologic responses over an acute 5-day period. Sustained severe inflammation, however, is associated with adverse outcomes.
One key question is how we might use biomarkers to distinguish physiologic from pathologic inflammation at a point that we can actually do something about it. I polled a number of my colleagues in regard to this question and received a variety of insightful answers. The most common response was “good question.” The answer is likely to be influenced by genotype and phenotype as well as acuity, severity, duration, disease or injury setting, and the degree of underlying malnutrition. Is there a tipping point that we can recognize beyond which inflammation becomes pathologic? There are many potential biomarkers that can be considered.57 Things may be too far along if we focus on downstream effects of severe and/or persistent inflammation. C-reactive protein, IL-6, IL-10, and procalcitonin offer potential opportunities. Associated immune dysfunction, including measures of ex vivo stimulation of TNF-α and HLA-DR markers on monocytes, may be an option. Decrease in albumin and associated expansion of the extravascular, extracellular fluid space are potential indicators. Markers of oxidative stress, micronutrient status, mitochondrial function, and organ injury are intriguing possibilities. Genetic predisposition is a potential indicator of inflammatory risk as a number of gene polymorphisms have been identified that appear to be associated with more robust inflammatory response57 and weight loss in cancer cachexia.58
Distinctive gene expression arrays have also been characterized in the setting of systemic inflammatory response.59-61 Last, the potential for systems biology approaches using metabolic pheno- typing is a very exciting recent development in biomarker assessment.59,62-65 A metabolomics approach can be used to predict early onset of systemic inflammatory response syndrome in patients with major trauma according to variation in levels of carbohydrate, amino acids, glucose, lactate, glutamine signals, and fatty acyl chains and lipids.62 Large-scale discovery proteomics using high-resolution liquid chromatography–mass spectrometry and multiplex cytokine analysis revealed a variety of candidate biomarker proteins that correlated well with survival in severe burn patients.63 Real-time surgical diagnostics with intraoperative tissue sampling directly linked to high-speed mass spectrometry and chemometrics have been proposed to guide surgeons in risk projection and treatment.64 Cancer-specific metabolic signatures have been identified for rapid diagnosis and staging of colorectal cancer using high-resolution magic angle spinning nuclear magnetic resonance (HR-MAS NMR).65
Opportunities for Intervention
A host of nutrition-related modulators of inflammation offer opportunities for intervention. They include ω-3 fatty acids, antioxidants, bioactive food components, anti-inflammatory diets, and caloric restriction.1 While a detailed discussion of these topics is beyond the scope of my Rhoads Lecture, it should be highlighted that the incorporation of an inflammatory component into our understanding of malnutrition establishes a construct for the use of anti-inflammatory interventions and sets the stage for nutrition practitioners to be integral members of the medical team. I envision trained nutrition professionals as guiding the medical team in appropriate nutrition assessment, diagnosis, and application of such therapies.
Conclusions
Inflammation and malnutrition are intimate partners that can be difficult to distinguish in clinical practice. We are just beginning to appreciate the complexities of their interactions. Our research agenda must address deficiencies in diagnostics, biomarkers, and therapeutics of inflammation in relation to malnutrition.18 Research to identify better indicators of both inflammation and malnutrition must be an especially high priority. “Omics” approaches have the potential to help us discern risk, guide interventions, and monitor response to therapies. Dissemination of the Academy–A.S.P.E.N. consensus recommended clinical characteristics for the diagnosis of malnutrition is ongoing. Education and training of allied practitioners in application of the new approach have been initiated. Supporting screening and assessment tools are in development. It is important to recognize that this is a work in progress. Some aspects are likely to serve us well; some are likely to evolve. Laboratory, functional, food intake, and body weight criteria warrant further testing. Translation of the proposed diagnostic approach to routine clinical practice will require validation. Inflammation can be a good thing; let’s try to keep it that way.
I would be remiss if I did not acknowledge the many contributions of colleagues and collaborators that culminated in the observations summarized in my Rhoads Lecture. There are too many to name, but I extend my most gracious thanks to all.
References
1. Jensen GL. Inflammation as the key interface of the medical and nutrition universes: a provocative examination of the future of nutrition and medicine. JPEN J Parenter Enteral Nutr. 2006;30(5):453-463.
2. Dudrick SJ, Wilmore DH, Vars HM, Rhoads JE. Can intravenous feeding as the sole means of nutrition support growth in the child and restore weight loss in an adult? An affirmative answer. Ann Surg. 1969;169(6):974-984.
3. Waterlow JC. Classification and definition of protein-calorie malnutrition. Br Med J. 1972;3:566-569.
4. Bistrian BR, Blackburn GL, Hallowell E, Heddle R. Protein status of general surgical patients. JAMA. 1974;230:858-860.
5. Bistrian BR, Blackburn GL, Vitale J, Cochran D, Naylor J. Prevalence of malnutrition in general medical patients. JAMA. 1976;235(15):1567-1570.
6. Butterworth CE. The skeleton in the hospital closet. Nutr Today. 1974;9:4-8.
7. Baker JP, Detsky AS, Wesson DE, et al. Nutritional assessment: a comparison of clinical judgment and objective measurements. N Engl J Med. 1982;306(16):969-972.
8. Detsky AS, McLaughlin JR, Baker JP, et al. What is subjective global assessment? JPEN J Parenter Enteral Nutr. 1987;11(1):8-13.
9. Leaning J, Guha-Sapir D. Global health: natural disasters, armed conflict, and public health. N Engl J Med. 2013;369:1836-1842.
10. Ledikwe JH, Smiciklas-Wright H, Mitchell DC, Miller CK, Jensen GL. Dietary patterns of rural older adults are associated with weight and nutritional status. J Am Geriatr Soc. 2004;52:589-595.
11. Correia Horvath JD, Dias de Castro ML, Kops N, Kruger Malinoski N, Friedman R. Obesity coexists with malnutrition? Adequacy of food consumption by severely obese patients to dietary reference intake recom- mendations. Nutr Hosp. 2014;29(2):292-299.
12. Leibovitz E, Giryes S, Makhline R, Zikri Ditch M, Berlovitz Y, Boaz M. Malnutrition risk in newly hospitalized overweight and obese individuals: Mr NOI. Eur J Clin Nutr. 2013;67(6):620-624.
13. Roubenoff R. Sarcopenic obesity: the confluence of two epidemics. Obes Res. 2004;12(6):887-888.
14. Stenholm S, Harris TB, Rantanen T, Visser M, Kritchevsky SB, Ferrucci L. Sarcopenic obesity—definition, etiology and consequences. Curr Opin Clin Nutr Metab Care. 2008;11(5):693-700.
15. Jensen GL, Hsiao PY. Obesity in older adults: relationship to functional limitation. Curr Opin Clin Nutr Metab Care. 2010;13:46-51.
16. Kohara K. Sarcopenic obesity in aging population: current status and future directions for research. Endocrine. 2014;45:15-25.
17. Overseas Development Institute. Overweight and obese adults reaching almost a billion in developing countries, as numbers continue to grow in richer nations. http://www.odi.org.uk/news/703-overweight-obese-adults- reaching-almost-billion-developing-countries-as-numbers-continue-grow-richer-nations. Accessed March 3, 2014.
18. Jensen GL, Bistrian B, Roubenoff R, Heimburger DC. Malnutrition syndromes: a conundrum vs continuum. JPEN J Parenter Enteral Nutr. 2009;33:710-716.
19. Corkins MR, Guenter P, Dimaria-Ghalili RA, et al.; American Society for Parenteral and Enteral Nutrition. Malnutrition diagnoses in hospitalized patients: United States, 2010 [published online November 18, 2013]. JPEN J Parenter Enteral Nutr.
20. Robinson MK, Trujillo EB, Mogensen KM, Rounds J, McManus K, Jacobs DO. Improving nutritional screening of hospitalized patients: the role of prealbumin. JPEN J Parenter Enteral Nutr. 2003;27:389-395.
21. Somanchi M, Tao X, Mullin GE. The facilitated early enteral and dietary management effectiveness trial in hospitalized patients with malnutrition. JPEN J Parenter Enteral Nutr. 2011;35:209-216.
22. Barker LA, Gout BS, Crowe TC. Hospital malnutrition: prevalence, identification and impact on patients and the healthcare system. Int J Environ Res Public Health. 2011;8(2):514-527.
23. Roubenoff R, Heymsfield SB, Kehayias JJ, Cannon JG, Rosenberg IH. Standardization of nomenclature of body composition in weight loss. Am J Clin Nutr. 1997;66:192-196.
24. Arends J, Bodoky G, Bozzetti F, et al. ESPEN guidelines on enteral nutrition: non-surgical oncology. Clin Nutr. 2006;25:245-259.
25. Evans WJ, Morley JE, Argilés J, et al. Cachexia: a new definition. Clin Nutr. 2008;27(6):793-799.
26. Muscaritoli M, Anker SD, Argilés J, et al. Consensus definition of sarcopenia, cachexia and pre-cachexia: joint document elaborated by Special Interest Groups (SIG) “cachexia-anorexia in chronic wasting diseases” and “nutrition in geriatrics.” Clin Nutr. 2010;29(2):154-159.
27. Fearon K, Strasser F, Anker SD, et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 2011;12(5):489-495.
28. Rosenburg IH. Epidemiologic and methodologic problems in determining nutritional status of older persons. Am J Clin Nutr. 1989;50(suppl):1231-1233.
29. Rosenberg IH. Sarcopenia: origins and clinical relevance. Clin Geriatr Med. 2011;27(3):337-339.
30. Delmonico MJ, Harris TB, Visser M, et al; Health, Aging, and Body. Longitudinal study of muscle strength, quality, and adipose tissue infiltra- tion. Am J Clin Nutr. 2009;90(6):1579-1585.
31. Cesari M, Fielding RA, Pahor M, et al; International Working Group on Sarcopenia. Biomarkers of sarcopenia in clinical trials-recommenda- tions from the International Working Group on Sarcopenia. J Cachexia Sarcopenia Muscle. 2012;3(3):181-190.
32. Fielding RA, Vellas B, Evans WJ, et al. Sarcopenia: an undiagnosed con- dition in older adults. Current consensus definition: prevalence, etiology, and consequences. International Working Group on Sarcopenia. J Am Med Dir Assoc. 2011;12:249-256.
33. Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al.; European Working Group on Sarcopenia in Older People. Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412-423.
34. Beyer I, Mets T, Bautmans I. Chronic low-grade inflammation and age-related sarcopenia. Curr Opin Clin Nutr Metab Care. 2012;15(1):12-22.
35. Batsis JA, Barre LK, Mackenzie TA, Pratt SI, Lopez-Jimenez F, Bartels SJ. Variation in the prevalence of sarcopenia and sarcopenic obesity in older adults associated with different research definitions: dual-energy X-ray absorptiometry data from the National Health and Nutrition Examination Survey 1999-2004. J Am Geriatr Soc. 2013;61(6):974-980.
36. Soeters PB, Reijven PLM, Van Bokhorst-de van der Schueren MAE, et al. A rational approach to nutritional assessment. Clin Nutr. 2008; 27:706- 716.
37. Jensen GL, Mirtallo J, Compher C, et al.; International Consensus Guideline Committee. Adult
starvation and disease-related malnutrition: a proposal for etiology-based diagnosis in the clinical practice setting from the International Consensus Guideline Committee. JPEN J Parenter Enteral Nutr. 2010;34(2):156-159.
38. Jensen GL, Mirtallo J, Compher C, et al; International Consensus Guideline Committee. Adult starvation and disease-related malnutrition: a proposal for etiology-based diagnosis in the clinical practice setting from the International Consensus Guideline Committee. Clin Nutr. 2010;29(2):151-153.
39. Prakash D. Anemia in the ICU: anemia of chronic disease versus anemia of acute illness. Crit Care Clin. 2012;28(3):333-343.
40. Gangat N, Wolanskyj AP. Anemia of chronic disease. Semin Hematol. 2013;50(3):232-238.
41. Gartner A, Berger J, Bour A, et al. Assessment of iron deficiency in the context of the obesity epidemic: importance of correcting serum ferritin concentrations for inflammation. Am J Clin Nutr. 2013;98(3):821-826.
42. Querfeld U. Vitamin D and inflammation. Pediatr Nephrol. 2013;28:605-610.
43. Waldron JL, Ashby HL, Cornes MP, et al. Vitamin D: a negative acute phase reactant. J Clin Pathol. 2013;66(7):620-622.
44. Ansemant T, Mahy S, Piroth C, et al. Severe hypovitaminosis D correlates with increased inflammatory markers in HIV infected patients. BMC Infect Dis. 2013;13:7.
45. Foster M, Samman S. Zinc and regulation of inflammatory cytokines: implications for cardiometabolic disease. Nutrients. 2012;4(7):676-694.
46. Chasapis CT, Loutsidou AC, Spiliopoulou CA, Stefanidou ME. Zinc and human health: an update. Arch Toxicol. 2012;86(4):521-534.
47. White JV, Guenter P, Jensen G, Malone A, Schofield M; Academy Malnutrition Work Group; A.S.P.E.N. Malnutrition Task Force; A.S.P.E.N. Board of Directors. Consensus statement: Academy of Nutrition and Dietetics and American Society for Parenteral and Enteral Nutrition: characteristics recommended for the identification and documentation of adult malnutrition (undernutrition). JPEN J Parenter Enteral Nutr. 2012;36(3):275-283.
48. White JV, Guenter P, Jensen G, Malone A, Schofield M; Academy of Nutrition and Dietetics Malnutrition Work Group; A.S.P.E.N. Malnutrition Task Force; A.S.P.E.N. Board of Directors. Consensus statement of the Academy of Nutrition and Dietetics/American Society for Parenteral and Enteral Nutrition: characteristics recommended for the identification and documentation of adult malnutrition (undernutrition). J Acad Nutr Diet. 2012;112(5):730-738.
49. Jensen GL, Hsiao PY, Wheeler D. Adult nutrition assessment tutorial. JPEN J Parenter Enteral Nutr. 2012;36:267-274.
50. Nicolo M, Compher CW, Still C, Huseini M, Dayton S, Jensen GL. Feasibility of accessing data in hospitalized patients to support diagnosis of malnutrition by the Academy-A.S.P.E.N. malnutrition consensus recommended clinical characteristics [published online December 3, 2013]. JPEN J Parenter Enteral Nutr.
51. Mehta NM, Corkins MR, Lyman B, et al; American Society for Parenteral and Enteral Nutrition Board of Directors. Defining pediatric malnutrition: a paradigm shift toward etiology-related definitions. JPEN J Parenter Enteral Nutr. 2013;37(4):460-481.
52. Cohen S, Janicki-Deverts D, Miller GE. Psychological stress and disease. JAMA. 2007;298(14):1685-1687.
53. Schultze-Florey CR, Martínez-Maza O, Magpantay L, et al. When grief makes you sick: bereavement induced systemic inflammation is a question of genotype. Brain Behav Immun. 2012;26(7):1066-1071.
54. Cohen S, Janicki-Deverts D, Doyle WJ, et al. Chronic stress, glucocorticoid receptor resistance, inflammation, and disease risk. Proc Natl Acad Sci U S A. 2012;109(16):5995-5999.
55. Parsons HA, Baracos VE, Dhillon N, Hong DS, Kurzrock R. Body composition, symptoms, and survival in advanced cancer patients referred to a phase I service. PLoS One. 2012;7(1):e29330.
56. Puthucheary ZA, Rawal J, McPhail M, et al. Acute skeletal muscle wasting in critical illness. JAMA. 2013;310(15):1591-1600.
57. Paoloni-Giacobino A, Grimble R, Pichard C. Genomic interactions with disease and nutrition. Clin Nutr. 2003;22(6):507-514.
58. Deans DA, Tan BH, Ross JA, et al. Cancer cachexia is associated with the IL10-1082 gene promoter polymorphism in patients with gastroesophageal malignancy. Am J Clin Nutr. 2009;89(4):1164-1167.
59. LaRosa SP, Opal SM. Biomarkers: the future. Crit Care Clin. 2011;27:407-419.
60. Xiao W, Mindrinos MN, Seok J, et al. Inflammation and Host Response to Injury Large-Scale Collaborative Research Program. A genomic storm in critically injured humans. J Exp Med. 2011;208(13):2581-2590.
61. ong HR. Genetics and genomics in pediatric septic shock. Crit Care Med. 2012;40:1618-1626.
62. Mao H, Wang H, Wang B, et al. Systemic metabolic changes of traumatic critically ill patients revealed by an NMR-based metabonomic approach. J Proteome Res. 2009;8(12):5423-5430.
63. Finnerty CC, Jeschke MG, Qian WJ, et al; Investigators of the Inflammation and the Host Response Glue Grant. Determination of burn patient outcome by large-scale quantitative discovery proteomics. Crit Care Med. 2013;41(6):1421-1434.
64. Nicholson JK, Holmes E, Kinross JM, Darzi AW, Takats Z, Lindon JC. Metabolic phenotyping in clinical and surgical environments. Nature. 2012;491(7424):384-392.
65. Mirnezami R, Jiménez B, Li JV, et al. Rapid diagnosis and staging of colorectal cancer via high-resolution magic angle spinning nuclear magnetic resonance (HR-MAS NMR) spectroscopy of intact tissue biopsies [published online July 15, 2013]. Ann Surg.
Financial disclosure: None declared.
This article originally appeared online
on April 7, 2014
Received for publication March 5, 2014.
Accepted for publication March 7, 2014.
.
Agradecimientos especiales a Gordon L. Jensen, M.D., Ph.D. Senior Associate Dean for Research Professor of Medicine and Nutrition The University of Vermont College of Medicine. Department of Nutritional Sciences, Penn State University (autor del artículo); Megan White. Publications Manager American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.); Michelle Binur. Rights Coordinator SAGE Publishing; Mrs. Addie Marie Bergeron Acanda por haber hecho posible la publicación de este importante trabajo en nuestra revista.